|
The calculation from the mass of Al to the mass of alum could be done in a single step ( Help Me). Actual yield is the amount of product obtained when the reaction is actually performed. Your result should be 35.1% to the correct number of significant figures, although this would often only reported as 35% ( Help Me). This is the theoretical yield. CAUTION! the molar mass of alum includes K, Al, S and O and the twelve H2O! You should get 8.351 g of alum ( Help Me).ĭetermine the percent yield. You should have found that the reaction could form 0.01750moles of alum ( Help Me).Ĭalculate the mass of alum (in grams) from moles of alum. Now convert moles of Al to moles of alum using the stoichiometric factor from the balanced chemical equation. You should get 0.01750 moles of Al ( Help Me). It gives you the easiest way to find the value of yield of a theoretical equation. Note that throughout this page extra insignificant figures carried along in the calculation to prevent rounding errors.įirst, we must determine the moles of Al in 0.475 g of Al. Percent yield calculator is a very useful tool for students of chemistry. Let’s assume that we used 0.475 g of Al and that we obtained 2.930 g of alum. is a calculation based simply on the overall balanced chemical equation. As per the percent yield formula, percent yield is equals to the actual yield, divided by theoretical yield and multiphy by 100. Since we do not need to determine the limiting reagent, our first step is to determine the amount of alum that can theoretically be formed from the amount of Al that we have. After the reaction is over, we add three more: actual yield, theoretical yield. The percentage yield calculator uses percent yield formula to show results. To simplify things we have told you that the Al is the limiting reagent (if you wish, you can check this). 
The percent yield is then simply the actual amount of product obtained divided by the theoretical yield times 100.įor the reaction of Al with KOH to form alum the balanced chemical reaction is as follows:Ģ Al (s) + 2 KOH (aq) + 22 H2O (l) + 4 H2SO4 (aq) → 2 KAl(SO4)2 + 12 H2O (s) + 3 H2 (g) From the balanced chemical reaction and the amount of reactants, we determine first the limiting reagent and then theoretical yield of the product. Insert required values into the given boxes and get accurate results in no time with percent yield calculator app.To determine the percent yield of a product in a chemical reaction we need to know the amount of all reactants used, the amount of the product formed and the balanced chemical reaction. Give a try this app to quickly calculate chem equations. I show you how to calculate percentage yield using a worked example and then give you a question to try yourself. 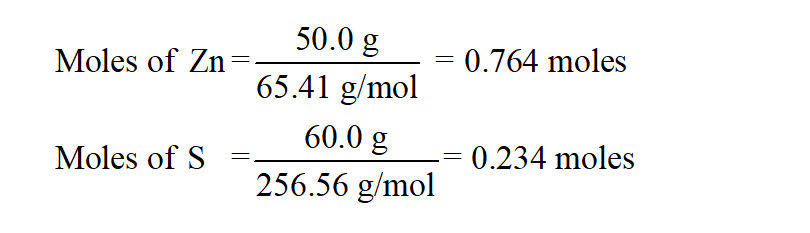
In this lesson you will learn how to calculate the perce. In this video we look at percentage yield. This tool makes your life easy with auto implementation of the percentage yield formula. This lesson is relevant for Grade 11, Grade 12 (Both IEB and DBE) and First year Chemistry Students. The percent of starting material that is converted to product in a chemical reaction is referred to as the percent yield. Because it is a simple and easy to use yield calculator. Apart from this, based on our calculation, we calculate the expected yield of. Automatically measure and analyze the fraction of actual and theoretical yield. It is the actual yield that we get after the completion of a chemical reaction. Get an accurate solution of percentage yield. This app provides you with the exact value of theoretical yield in a single tap. Other calculators who analyze the reaction of chemistry equations also do not have this feature to calculate the percent yield with reaction. Theoretical yield is the yield that is calculated. But this calculator enables you to calculate yield values. In chemistry, percentage yield is the ratio of the actual yield to the theoretical yield multiplied by 100. There are many apps in the market which help you to find the fraction of any equation. If you want to calculate the percentage yield of any theoretical problem! Insert the required values into the empty fields and tap on the calculate button to get accurate results with this yield calculator. See the following formula: Wrapping It Up Across the field of chemistry, it is important to be able to calculate percent yield quickly and easily. It is easy to calculate the theoretical yield percentage with this calculator. To calculate percent yield, simply divide the actual yield by the theoretical yield and multiply by 100. 
No need to do manual calculations with yield formulas when you have this calculator. Percent yield calculator is a very useful tool for students of chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
